2 It is then convenient convenient in the absence of further information) to take that common value to be zero: S (0) 0. The second, based on the fact that entropy is a state function, uses a thermodynamic cycle similar to those discussed previously. That will be the change in entropy for any process going from state 1 to state 2. This is one definition used for the arrow of time since entropy of the universe will always increase over time according to the second law of thermodynamics. The third law of thermodynamics is an assertion about the unattainability of absolute zero, but it implies that the entropies of all pure, perfectly crystalline substances are the same at T 0. The first, based on the definition of absolute entropy provided by the third law of thermodynamics, uses tabulated values of absolute entropies of substances. We just need to find or imagine a reversible process that takes us from state 1 to state 2 and calculate \(\Delta S\) for that process. Thus the change in entropy \(\Delta S\) of a system between state 1 and state 2 is the same no matter how the change occurs. Temperature is defined via entropy: The inverse of temperature (1/T) is defined as the change of entropy (S) in a system, when the total energy E of the system. Entropy is a key concept in physics and chemistry, with application in other disciplines, including cosmology, biology, and economics. The reason is that the entropy \(S\) of a system, like internal energy \(U\) depends only on the state of the system and not how it reached that condition. However, we can find \(\Delta S\) precisely even for real, irreversible processes. Zero kelvin (273.15 ☌) is defined as absolute zero. The definition of \(\Delta S\) is strictly valid only for reversible processes, such as used in a Carnot engine. The units of entropy change are cal/K/mol in CGS system and joules/K/mol in. Entropy change during a process is defined as the amount of heat ( q ) absorbed isothermally and reversibly divided by the absolute Temperature ( T ) at which the heat is absorbed. If temperature changes during the process, then it is usually a good approximation (for small changes in temperature) to take \(T\) to be the average temperature, avoiding the need to use integral calculus to find \(\Delta S\). This shows that entropy change is inversely proportional to temperature. 1 mol of NH 3 (g) or 1 mol of He (g), both at 25☌. Predict which substance in each pair has the higher entropy and justify your answer. The SI unit for entropy is joules per kelvin (J/K). The magnitude of the increase is greater than the magnitude of the decrease, so the overall entropy change for the formation of an NaCl solution is positive. Which means that how much heat is removed from the body at lower temperature. Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other. 
Where \(Q\) is the heat transfer, which is positive for heat transfer into and negative for heat transfer out of, and \(T\) is the absolute temperature at which the reversible process takes place. Absolute Entropy, Unattainability of Absolute Zero) And Its Limitation. Entropy is a state function that is often erroneously referred to as the state of disorder of a system. In this case, the reaction is highly exothermic, and the drive towards a decrease in energy allows the reaction to occur.,\] According to the drive towards higher entropy, the formation of water from hydrogen and oxygen is an unfavorable reaction. Much as the ideal gas temperature scale has a natural zero at the temperature at which the volume extrapolates to zero, a perfect crystalline substance has a natural. Calculate entropy changes for phase transitions and chemical reactions under standard conditions. By the Lewis and Randall statement of the third law, the entropy of a substance that forms a perfect crystal is identically equal to zero at absolute zero. For this case, the probability of each microstate of the system is equal, so it was equivalent. For example, a glassy solid and a solid containing a mixture of isotopes. The limitation of this law is that many solids do not have zero entropy at absolute zero. It is because there is a perfect order in a crystal at absolute zero. 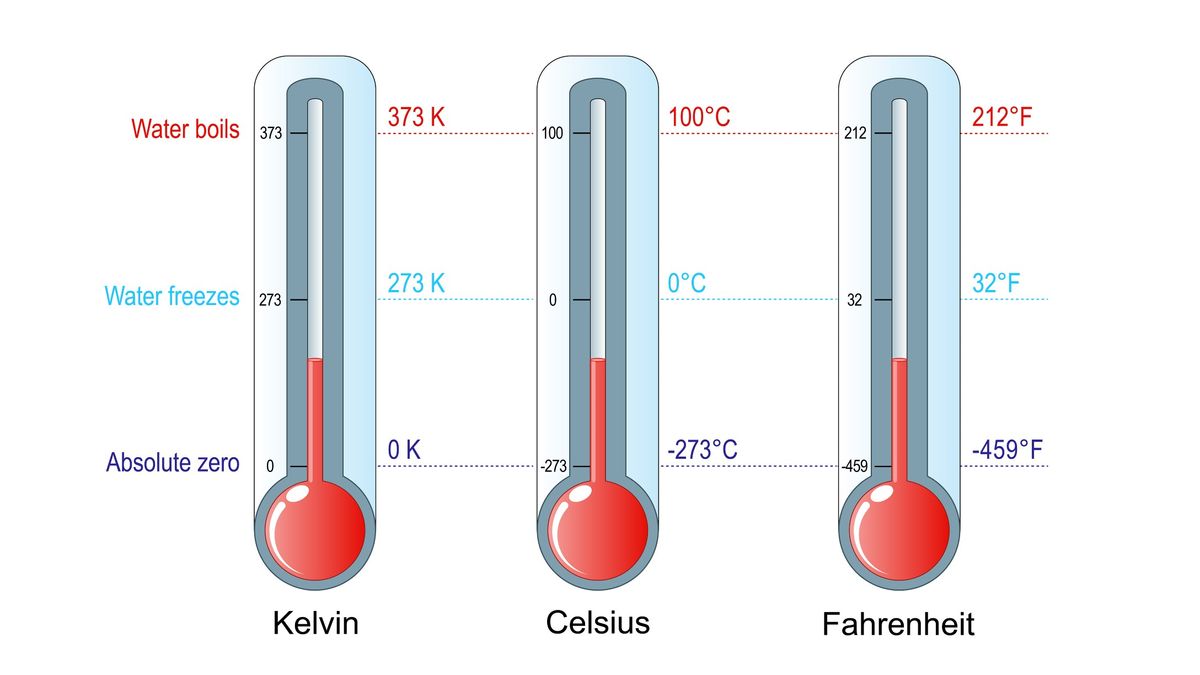
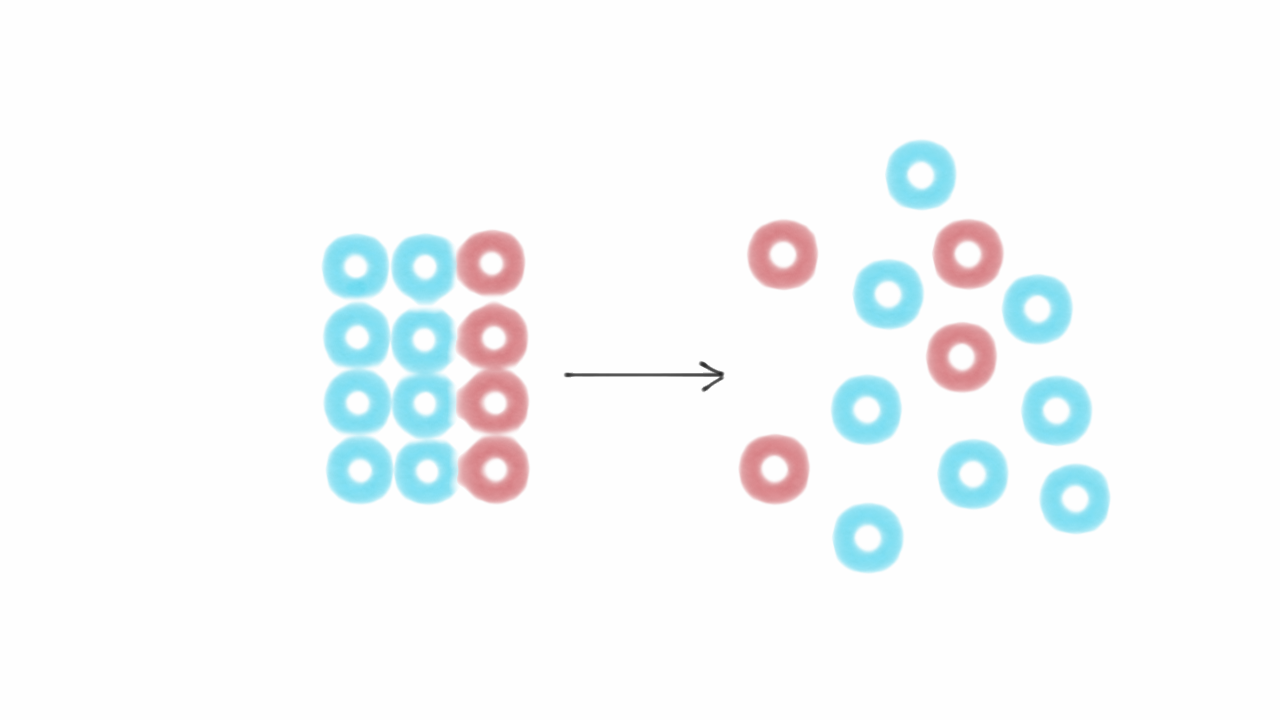
Boltzmann's paradigm was an ideal gas of N identical particles, of which Ni are in the i -th microscopic condition (range) of position and momentum. The entropy of any crystalline solid approaches zero as the temperature approaches absolute temperature. The entropy change for this reaction is highly negative because three gaseous molecules are being converted into two liquid molecules. The absolute entropy of a pure substance at a given temperature is the sum of all the entropy it would acquire on warming from absolute zero (where S 0. Interpreted in this way, Boltzmann's formula is the most basic formula for the thermodynamic entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
